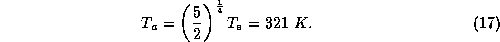Lecture 2: Physics of the Tropical Atmosphere, I



Next: Lecture 3: Physics
Up: Tropical Cyclones
Previous: Lecture 1: Observed
To understand the origin of tropical cyclones, it is necessary to begin with a
discussion of the physics underlying the normal state of the tropical maritime
atmosphere. The average vertical thermal structure of the atmosphere is
illustrated in Figure 2.1. The lowest layer, the troposphere, extends from the
surface upward to 8-15 km, depending mostly on the latitude and season. It is
characterized by temperature decreasing with altitude, at an average rate of
about 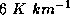 . This upward decrease of temperature ends, often
abruptly, at the tropopause, above which the temperature is constant or
increases with altitude. This layer is called the stratosphere. At an altitude
of between 40 and 50 km., the temperature lapse rate again changes sign, at the
stratopause, and in the mesosphere the temperature decreases upward. Above the
mesopause lies the thermosphere, in which the temperature once again increases
with altitude, and here the mean free path between molecular collisions becomes
appreciable. The high temperatures of the upper thermosphere are caused by
collisions between atmospheric constituents and high energy particles
originating in the solar wind; the actual temperature of this region is a strong
function of the level of solar substorm activity. But below the thermosphere,
the temperature is determined by transfer of electromagnetic radiation, and in
the troposphere convective heat transfer is important as well.
. This upward decrease of temperature ends, often
abruptly, at the tropopause, above which the temperature is constant or
increases with altitude. This layer is called the stratosphere. At an altitude
of between 40 and 50 km., the temperature lapse rate again changes sign, at the
stratopause, and in the mesosphere the temperature decreases upward. Above the
mesopause lies the thermosphere, in which the temperature once again increases
with altitude, and here the mean free path between molecular collisions becomes
appreciable. The high temperatures of the upper thermosphere are caused by
collisions between atmospheric constituents and high energy particles
originating in the solar wind; the actual temperature of this region is a strong
function of the level of solar substorm activity. But below the thermosphere,
the temperature is determined by transfer of electromagnetic radiation, and in
the troposphere convective heat transfer is important as well.
The two major constituents of the atmosphere, molecular oxygen and nitrogen, are
very nearly transparent to both solar and terrestrial radiation, although some
scattering of solar radiation does occur. Oxygen and nitrogen are diatomic
molecules with a very limited number of degrees of freedom, thus they are poor
absorbers of radiation. If these were the only gases in the atmosphere, then it
would be almost completely transparent and the earth's surface would radiate at
very nearly the black body temperature necessary to balance the input of solar
radiation. Let's calculate what this temperature would be. At the mean radius
of the earth's orbit about the sun, the flux of solar radiation, 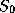 , is known
as the solar constant and has a value of about
, is known
as the solar constant and has a value of about 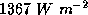 . By
elementary geometry, the amount of this radiation intercepted by the earth, per
unit surface area of the earth, is just
. By
elementary geometry, the amount of this radiation intercepted by the earth, per
unit surface area of the earth, is just 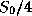 . A fraction of this radiation,
. A fraction of this radiation,
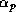 is reflected back to space by the earth's surface and by clouds; this
fraction is known as the planetary albedo. By the Stefan-Boltzmann law,
the black body temperature is given by
is reflected back to space by the earth's surface and by clouds; this
fraction is known as the planetary albedo. By the Stefan-Boltzmann law,
the black body temperature is given by
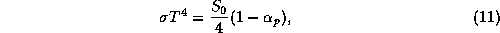
where  is
is 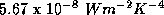 . Using a
planetary albedo of 0.3, this gives a black body temperature of 255 K or -18 C,
very much colder than the average surface temperature of the earth, which is 288
K or 15 C.
. Using a
planetary albedo of 0.3, this gives a black body temperature of 255 K or -18 C,
very much colder than the average surface temperature of the earth, which is 288
K or 15 C.
The surface temperature of the earth is higher than its black body temperature
owing to the presence of a few principally triatomic species, notably water
vapor, carbon dioxide and ozone. While these represent very small fractions of
the atmosphere by mass, they are critical absorbers and emitters of radiation,
because their structure allows for many more degrees of freedom; especially in
rotation and vibration. Condensed water, in the form of clouds, is also very
important in reflecting, absorbing and emitting radiation.
Figure 2.2 shows two black body curves, for the sun and earth, respectively, as
well as atmospheric absorption characteristics near the surface and near 11 km.
First note that, because the earth's effective emitting temperature is about 255
K whereas that of the sun is close to 6000 K, their two spectra have almost no
overlap. So we may usefully talk about solar and terrestrial radiation, or
shortwave and longwave radiation, without ambiguity. Also note that the solar
spectrum peaks in a region in which there is very little atmospheric absorption,
so that most sunlight that is not reflected or absorbed by clouds reaches the
surface. Virtually all of the very high energy, ultraviolet radiation is
absorbed in the middle stratosphere in a photochemical reaction in which
molecular oxygen breaks into 2 oxygen atoms, some of which combine with other
available oxygen atoms to form ozone (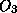 ). Once formed, ozone itself absorbs
some ultraviolet radiation. Were most of the incoming ultraviolet spectrum to
reach the surface, life as we know it would not be possible. The high
temperatures of the upper stratosphere are owing to the absorption of
ultraviolet radiation by ozone.
). Once formed, ozone itself absorbs
some ultraviolet radiation. Were most of the incoming ultraviolet spectrum to
reach the surface, life as we know it would not be possible. The high
temperatures of the upper stratosphere are owing to the absorption of
ultraviolet radiation by ozone.
In the terrestrial spectrum, there are large numbers of absorption lines (which
are broadened by both Doppler and pressure effects), owing mostly to the
triatomic molecules water vapor and carbon dioxide, but with some notable
contributions from other trace species such as methane. Water vapor content
decreases sharply with height, but there are still large water absorption
effects at 11 km. Taken together, the relative opacity of the atmosphere to
terrestrial radiation makes for a strong ``greenhouse effect".
The greenhouse effect is central to understanding why the tropical atmosphere
supports tropical cyclones. In particular, it necessitates a degree of
thermodynamic disequilibrium between the ocean and atmosphere. It is this
disequilibrium which supplies energy to tropical cyclones.
To understand the fundamental point, let's have a look at a very simple model of
the greenhouse effect, as shown in Figure 2.3. In the first instance, consider
2 layers of gas each with an emissivity of unity in the terrestrial band but
which are completely transparent to solar radiation. They overly a surface of
emissivity unity. In radiative equilibrium, each layer emits upward and
downward, and the surface emits upward. The energy balance for the upper layer
is
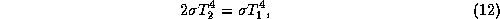
where 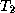 is the temperature of the upper layer and
is the temperature of the upper layer and 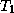 is the
temperature of the lower layer. Likewise, the energy balance of the lower layer
is given by
is the
temperature of the lower layer. Likewise, the energy balance of the lower layer
is given by
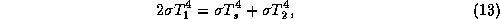
where 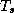 is the surface temperature. The surface energy balance is
simply
is the surface temperature. The surface energy balance is
simply
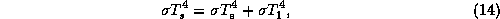
where 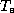 is the effective blackbody emission temperature, defined
is the effective blackbody emission temperature, defined

Now we may solve (12) - (14) for the three temperatures,
giving

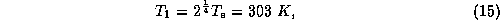

The surface temperature is substantially greater than the blackbody equilibrium
temperature. Note also that since 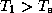 , the surface actually received more
radiation from the atmosphere than it does from the sun! More to the point, we
can show that the air in immediate contact with the ground is not in thermal
equilibrium with the ground. We can do this by inserting a vanishingly thin
layer of air between the surface and the first, opaque layer. This thin layer
has vanishing emissivity,
, the surface actually received more
radiation from the atmosphere than it does from the sun! More to the point, we
can show that the air in immediate contact with the ground is not in thermal
equilibrium with the ground. We can do this by inserting a vanishingly thin
layer of air between the surface and the first, opaque layer. This thin layer
has vanishing emissivity,  , and its energy balance is
, and its energy balance is
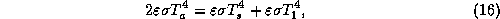
where 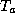 is the temperature of the thin layer next to the surface.
Since the layer is vanishingly thin, it does not affect the energy balance of
the surface and other layers. The solution to (16) is then
is the temperature of the thin layer next to the surface.
Since the layer is vanishingly thin, it does not affect the energy balance of
the surface and other layers. The solution to (16) is then
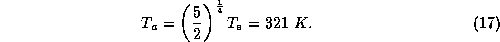
So clearly, in radiative equilibrium, the surface air is not in thermal
equilibrium with the surface. This creates the potential for convective
heat transport away from the surface. This will be the subject of the next
lecture.



Next: Lecture 3: Physics
Up: Tropical Cyclones
Previous: Lecture 1: Observed
Kerry Emanuel
Mon Apr 13 10:50:48 EDT 1998
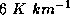 . This upward decrease of temperature ends, often
abruptly, at the tropopause, above which the temperature is constant or
increases with altitude. This layer is called the stratosphere. At an altitude
of between 40 and 50 km., the temperature lapse rate again changes sign, at the
stratopause, and in the mesosphere the temperature decreases upward. Above the
mesopause lies the thermosphere, in which the temperature once again increases
with altitude, and here the mean free path between molecular collisions becomes
appreciable. The high temperatures of the upper thermosphere are caused by
collisions between atmospheric constituents and high energy particles
originating in the solar wind; the actual temperature of this region is a strong
function of the level of solar substorm activity. But below the thermosphere,
the temperature is determined by transfer of electromagnetic radiation, and in
the troposphere convective heat transfer is important as well.
. This upward decrease of temperature ends, often
abruptly, at the tropopause, above which the temperature is constant or
increases with altitude. This layer is called the stratosphere. At an altitude
of between 40 and 50 km., the temperature lapse rate again changes sign, at the
stratopause, and in the mesosphere the temperature decreases upward. Above the
mesopause lies the thermosphere, in which the temperature once again increases
with altitude, and here the mean free path between molecular collisions becomes
appreciable. The high temperatures of the upper thermosphere are caused by
collisions between atmospheric constituents and high energy particles
originating in the solar wind; the actual temperature of this region is a strong
function of the level of solar substorm activity. But below the thermosphere,
the temperature is determined by transfer of electromagnetic radiation, and in
the troposphere convective heat transfer is important as well.
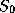 , is known
as the solar constant and has a value of about
, is known
as the solar constant and has a value of about 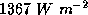 . By
elementary geometry, the amount of this radiation intercepted by the earth, per
unit surface area of the earth, is just
. By
elementary geometry, the amount of this radiation intercepted by the earth, per
unit surface area of the earth, is just 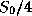 . A fraction of this radiation,
. A fraction of this radiation,
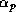 is reflected back to space by the earth's surface and by clouds; this
fraction is known as the planetary albedo. By the Stefan-Boltzmann law,
the black body temperature is given by
is reflected back to space by the earth's surface and by clouds; this
fraction is known as the planetary albedo. By the Stefan-Boltzmann law,
the black body temperature is given by
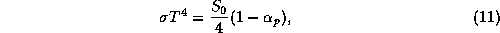
 is
is 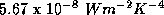 . Using a
planetary albedo of 0.3, this gives a black body temperature of 255 K or -18 C,
very much colder than the average surface temperature of the earth, which is 288
K or 15 C.
. Using a
planetary albedo of 0.3, this gives a black body temperature of 255 K or -18 C,
very much colder than the average surface temperature of the earth, which is 288
K or 15 C.
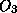 ). Once formed, ozone itself absorbs
some ultraviolet radiation. Were most of the incoming ultraviolet spectrum to
reach the surface, life as we know it would not be possible. The high
temperatures of the upper stratosphere are owing to the absorption of
ultraviolet radiation by ozone.
). Once formed, ozone itself absorbs
some ultraviolet radiation. Were most of the incoming ultraviolet spectrum to
reach the surface, life as we know it would not be possible. The high
temperatures of the upper stratosphere are owing to the absorption of
ultraviolet radiation by ozone.
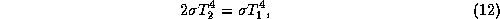
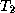 is the temperature of the upper layer and
is the temperature of the upper layer and 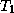 is the
temperature of the lower layer. Likewise, the energy balance of the lower layer
is given by
is the
temperature of the lower layer. Likewise, the energy balance of the lower layer
is given by
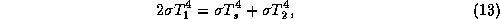
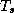 is the surface temperature. The surface energy balance is
simply
is the surface temperature. The surface energy balance is
simply
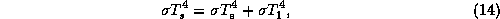
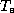 is the effective blackbody emission temperature, defined
is the effective blackbody emission temperature, defined


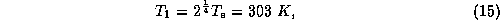

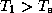 , the surface actually received more
radiation from the atmosphere than it does from the sun! More to the point, we
can show that the air in immediate contact with the ground is not in thermal
equilibrium with the ground. We can do this by inserting a vanishingly thin
layer of air between the surface and the first, opaque layer. This thin layer
has vanishing emissivity,
, the surface actually received more
radiation from the atmosphere than it does from the sun! More to the point, we
can show that the air in immediate contact with the ground is not in thermal
equilibrium with the ground. We can do this by inserting a vanishingly thin
layer of air between the surface and the first, opaque layer. This thin layer
has vanishing emissivity,  , and its energy balance is
, and its energy balance is
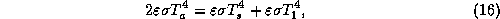
 is the temperature of the thin layer next to the surface.
Since the layer is vanishingly thin, it does not affect the energy balance of
the surface and other layers. The solution to (
is the temperature of the thin layer next to the surface.
Since the layer is vanishingly thin, it does not affect the energy balance of
the surface and other layers. The solution to (